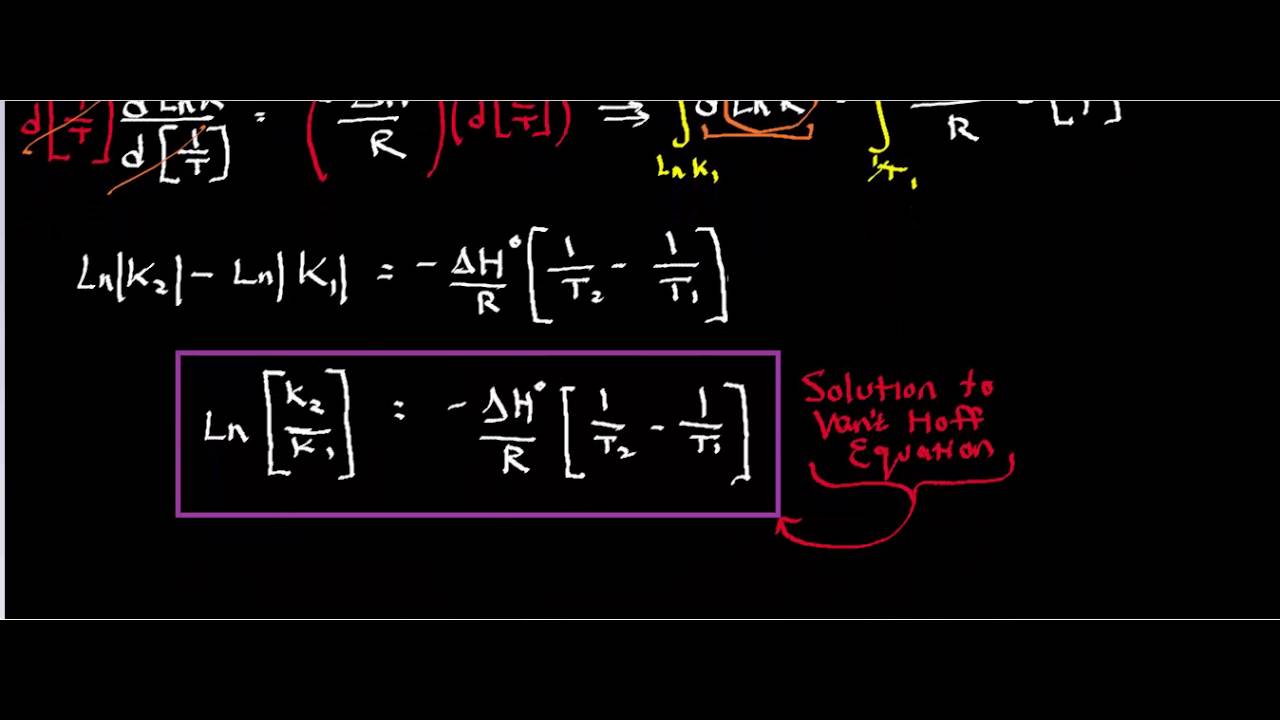
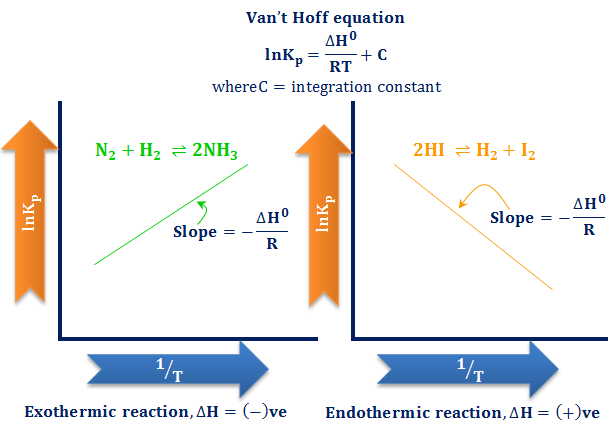
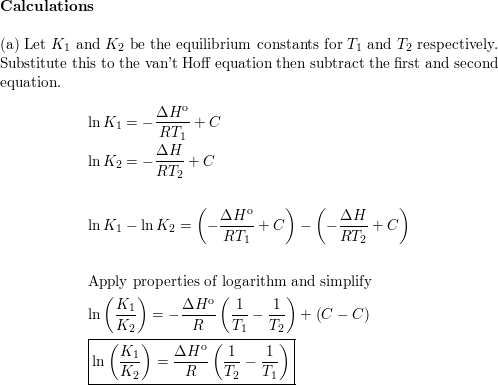The van't Hoff Equilibrium Box is here depicted at the beginning of... | Download Scientific Diagram
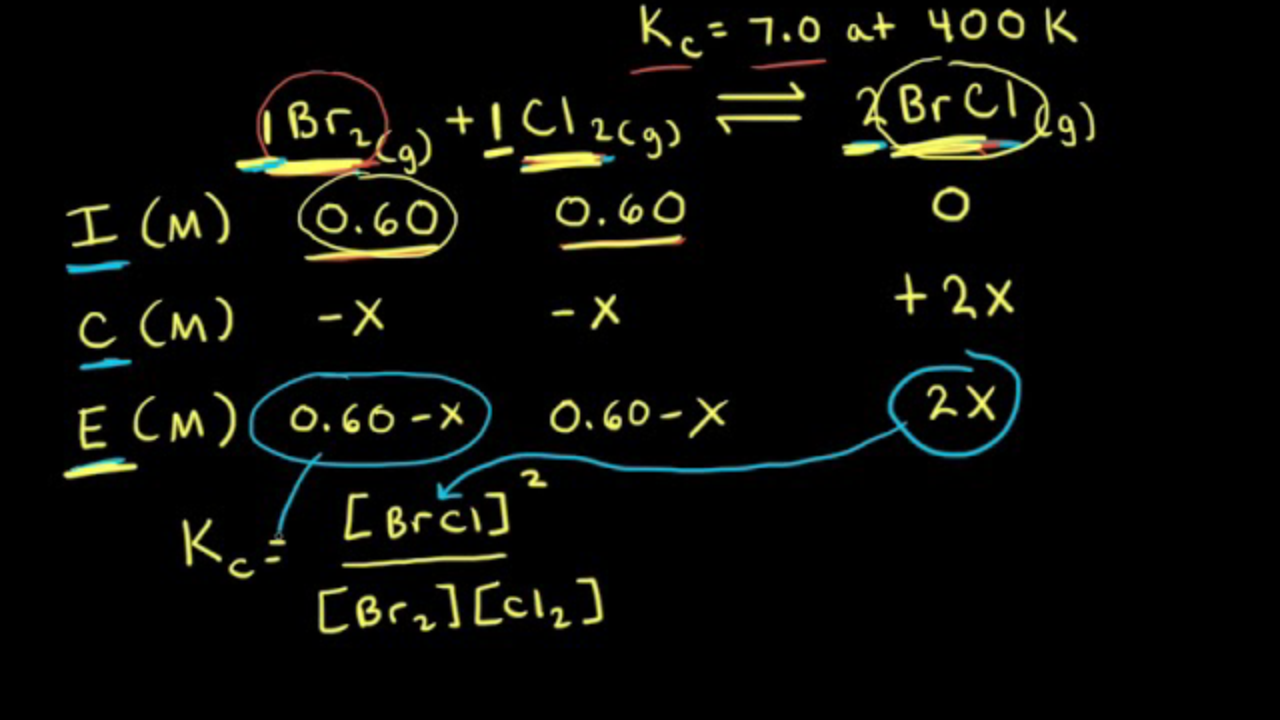
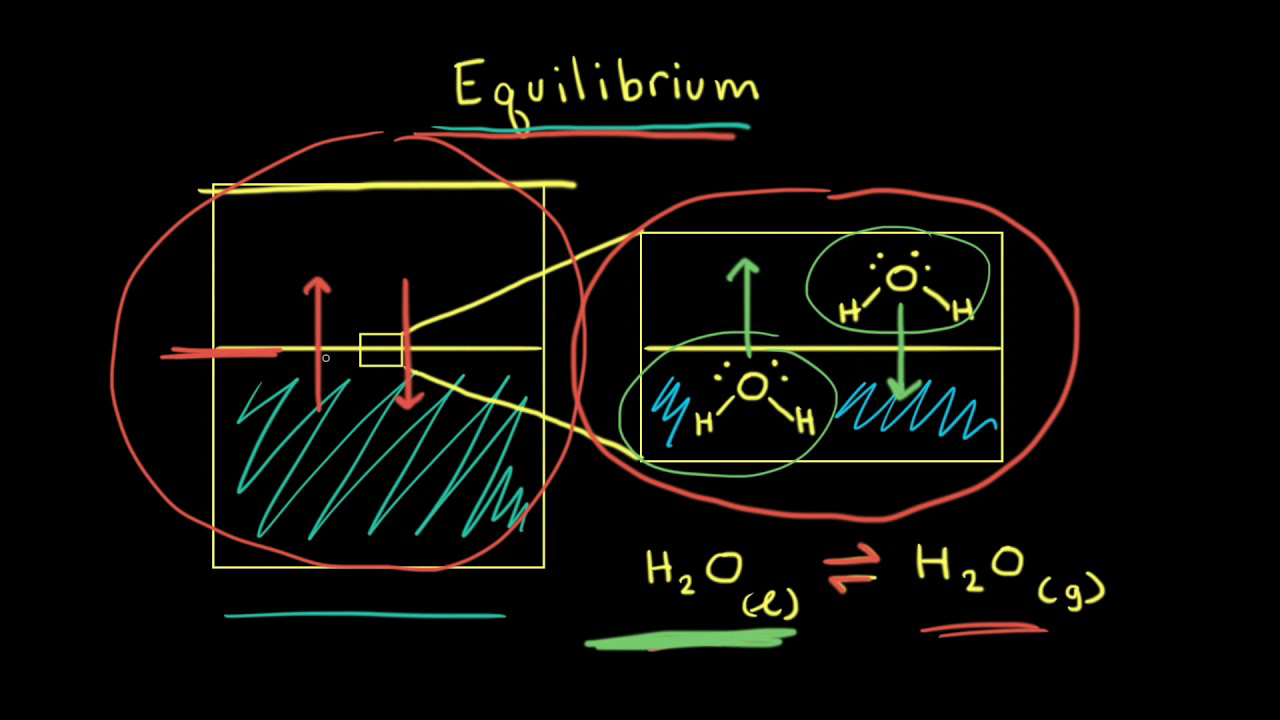
Calculating equilibrium concentrations from initial concentrations and the equilibrium constant (worked example) (video) | Khan Academy

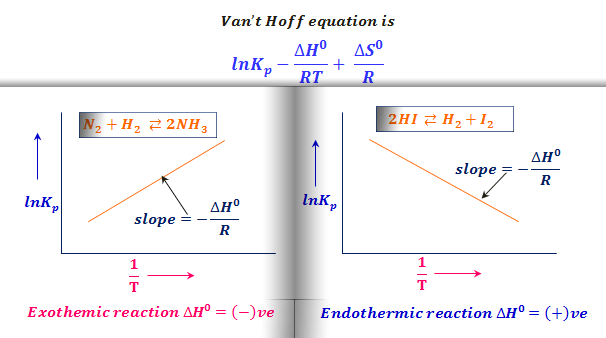
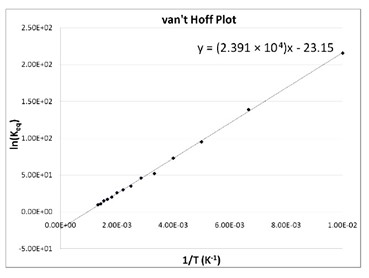
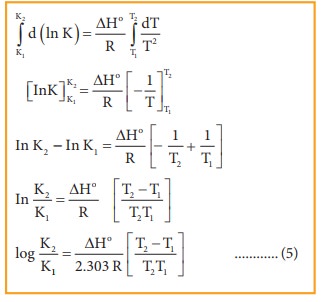
Example (Van't Hoff Equation) | 11th Chemistry | Physical and Chemical Equilibrium | தமிழ் | Part 23 - YouTube
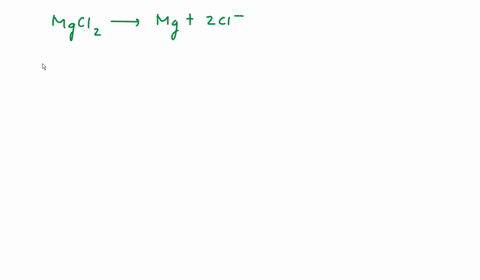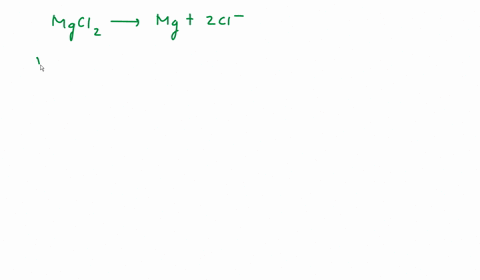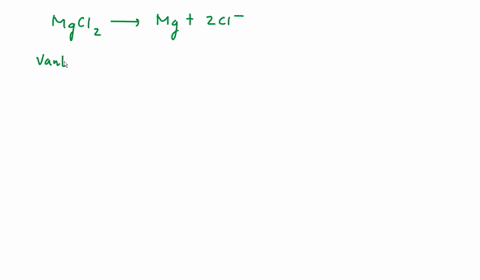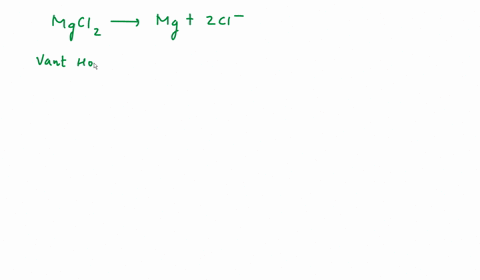
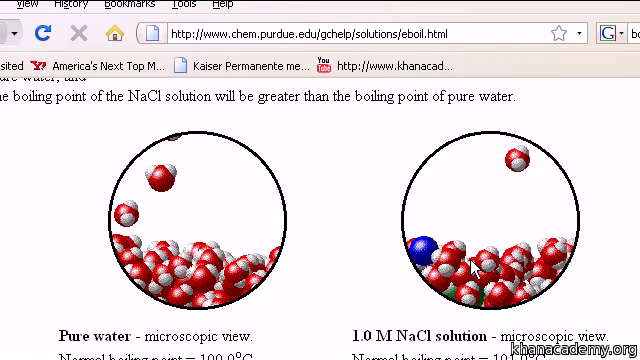
SOLVED: We often assume that the van't Hoff factor is simply equal to the number of ions present when the electrolyte dissociates in solution. In reality, van't Hoff factors are generally smaller